(via TheNewswire)
Calgary, Alberta, Canada – TheNewswire – March 16, 2026 - Voyageur Pharmaceuticals Ltd . ( TSX-V: VM ) (OTC Pink: VYYRF) (the "Company" or "Voyageur"), a pharmaceutical company focused on developing barium and iodine contrast media drugs for medical imaging, is pleased to present a corporate update and strategic roadmap to investors.
Voyageur aims to address the critical supply chain vulnerabilities in the diagnostic imaging sector, where demand is projected to grow from US$6.77 billion in 2024 to US$13.86 billion by 2033 ( Research Contrast ). With barite classified as a critical mineral by the U.S. Geological Survey, Voyageur's strategy involves creating a secure, reliable, and vertically integrated supply chain for both barium and iodine active pharmaceutical ingredients, in order to manufacture its barium and iodine contrast media drugs. This includes the entire value chain from mineral discovery to the manufacturing of finished products to ensure supply security and competitive production costs.
Voyageur is committed to strengthening healthcare infrastructure and meeting the growing global demand for diagnostic imaging pharmaceuticals. To support this strategy, the Company is advancing a disciplined and well-defined development pathway toward commercial-scale production of both barium and iodine contrast agents. Recognizing that the barium and iodine markets operate through distinct supply chains, Voyageur plans to establish a comprehensive integrated framework to support the manufacturing and distribution of a full portfolio of barium and iodine radiology contrast media drugs.
Voyageur is currently focused on completing two bankable feasibility studies (each a BFS ) on the following projects that underpin this vertically integrated strategy:
Voyageur Radiology Iodine Barium Drug Manufacturing Project
Bayer Iodine Project
These projects represent foundational steps toward establishing a resilient, North American–based supply chain capable of supporting the growing global demand for diagnostic imaging pharmaceuticals.
Voyageur Barium Strategy, Frances Creek FDA Barium/Iodine Approval Update:
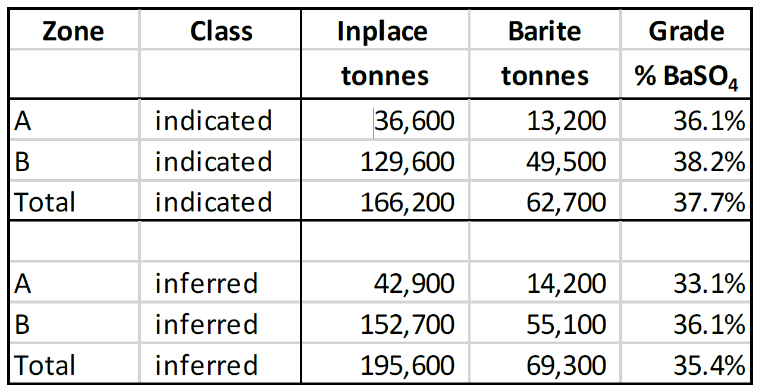
Voyageur is actively advancing the Frances Creek ( FC ) barium contrast project, now in the BFS phase. The project hosts an indicated and inferred mineral resource of 132,000 tonnes of pharmaceutical grade barium sulfate, forming a cornerstone of the Company’s strategy to establish a fully vertically integrated barium contrast media supply chain. Recent testing confirms the FC barite purity exceeds Pharmaceutical Grade (97.5%), with an average grade 98.8% BaSO 4 (for a description of the methodology used to determine the mineral resources, an explanation of how samples were collected, processed, and analyzed and a summary of the QA/QC (quality assurance/quality control) procedures applied see news release dated March 4, 2026 ).
Voyageur has completed product development initiatives for five Health Canada approved barium products with Alberta Veterinary Laboratories, a GMP pharmaceutical manufacturer, to produce barium contrast products using imported barium sulfate. Sales of these products are being incorporated into the ongoing FC barium BFS, which the Company expects to deliver in the second half of 2026, following technical requirements for the geotechnical drilling study at the FC quarry site.
Voyageur has generated approximately C$32,000 in initial contrast product sales, establishing commercial validation. Sales and pricing data across Canadian and international markets are a key contributor in demonstrating the manufacturing advantages and margin potential of Voyageur’s vertically integrated FC barium project. The Company completed a preliminary economic assessment that valued the FC barium contrast project , with a NPV of $344M (see news release dated January 11, 2022 ).
To enhance the FC BFS and future sales, Voyageur is building a strong network of clients in Canada, while developing international sales through clinics and distribution opportunities in Latin America, Middle East, North Africa, and Asia-Pacific regions. These efforts provide crucial market intelligence on pricing and cost dynamics amid supply shortages, while also identifying favorable conditions for distribution agreements and regulatory registrations, enabling Voyageur to meet urgent healthcare demands and expand global access to its barium contrast products. Voyageur is working towards having multiple customers in place for the launch of FC barium contrast production after securing the CapEx to build the barium and iodine drug manufacturing facility.
FDA
Regulatory pathways for barium and iodine contrast media are expected to follow an 18 to 24-month FDA review and approval process, potentially enabling U.S. barium market entry as soon as mid 2027 to late 2027. FDA approval, recognized globally, will also allow for registration of products in the European Union and other international markets, expanding Voyageur’s global growth opportunities. Domestic and international market data is being leveraged in the pending BFS to demonstrate the strategic and economic benefits of vertically integrating barium and iodine contrast drug production.
Voyageur is strategically advancing its U.S. regulatory initiatives to expand its presence in critical diagnostic imaging markets. The Company has completed the first drafts of all barium FDA submission documents and formally commenced the approval process in February 2026 via the 505(b)(2) regulatory pathway , positioning Voyageur to bring this essential diagnostic agent to market efficiently. The FDA is expected to provide all pending requirements in Q1 2026, after which the team will execute a comprehensive and detailed submission . The review process is anticipated to take approximately one year , with product testing projected at six to seven months , and the 505(b)(2) pathway, a hybrid U.S. New Drug Application (NDA) review, typically following a ten-month evaluation period . Building on this momentum, Voyageur plans to advance a generic iodine FDA license in the second half of 2026 , further strengthening its pharmaceutical portfolio and reinforcing its commitment to addressing critical medical supply needs.
Voyageur Iodine Drug Feasibility
Voyageur is independently advancing a prefeasibility study for iodine contrast media drug production , integrating the Mueller iodine extraction process with the Streamline iodine drug manufacturing platform . The project leverages iodine-rich brine sourced from oil and gas operations and water disposal companies in the United States, targeting 35 million doses per year of iodine contrast drug production . The Mueller process is focused on iodine extraction from Brine water, and the Streamline system is focused on creating iodine drugs from the iodine extracted from the water. These two processes combined, are expected to lower costs to manufacture iodine drugs significantly.
2. Bayer Iodine Project – US$2.35M Funding
Voyageur’s iodine strategy is also anchored by the Bayer Iodine production project, which focuses on completing a BFS. Subject to successful completion of the Bayer BFS, Bayer and Voyageur may consider advancing the project into a second phase, under an offtake-linked production financing arrangement. In this potential second phase, Bayer would provide capital financing for the project, while Voyageur would operate and manage the project. This collaboration, which began with the signing of an LOI with Bayer on December 20, 2024, represents an important early step in Voyageur's mission to become the first domestic producer of iodine contrast in the United States of America, while helping to secure critical supply chains for North America's healthcare industry. All intellectual property developed in connection with the project will remain exclusively owned by Voyageur.
Final Project Feasibility
Voyageur is engaging with a global engineering company, to complete the final two BFS for:
Voyageur radiology drug manufacturing project, to manufacture barium and iodine contrast. The FC BFS will be combined with Voyageur’s iodine drug prefeasibility study, which is in the final drafting phase, to create the final BFS for large scale contrast media drug production of both barium contrast and iodine contrast drugs.
Bayer Iodine project, to manufacture iodine feedstock for Bayer to use to manufacture iodine-based drugs.
The Company is on track to deliver a BFS for each of the two projects, positioning Voyageur to establish a secure, large-scale, North American supply of essential contrast media drugs.
Upon completion of the two BFS, Voyageur will have finalized the economics and plant design to vertically produce iodine and barium contrast media drugs From Earth to Bottle , becoming the only vertically integrated radiology drug company in the marketplace, with the potential added value from a non dilutive financing to produce iodine for Bayer. Voyageur is actively reviewing its financial options to fund future capital expenditures (CAPEX), targeting funding based on the finalized feasibility studies and plant construction for the Bayer project and for Voyageur’s barium and iodine contrast pharmaceutical manufacturing, targeting construction to commence in 2027.
Every initiative underway, from project development and regulatory approvals to strategic partnerships and process innovation, is being executed to maximize value for all stakeholders. Management of the Company believes these efforts will position Voyageur as the only vertically integrated manufacturer of barium and iodine contrast media globally, providing affordable and secure supplies for healthcare authorities worldwide and reinforcing the Company’s value proposition: From Earth to Bottle .
This disclosure has been reviewed and approved by Bradley Willis, P.Eng, a non-independent (by virtue of being a director and Chief Operating Officer of the Company) Qualified Person as defined by National Instrument 43-101 - Standards of Disclosure for Mineral Projects.
About Voyageur Pharmaceuticals Ltd.
Voyageur, a Canadian public company trading under the symbol VM on the TSXV, is in development of barium and iodine Active Pharmaceutical Ingredients (API) and intends to offer high-performance, cost-effective imaging contrast agents. With a strategic focus on vertically integrating the barium and iodine contrast markets, Voyageur aims to become a key player by producing its own barium, iodine, and new endohedral fullerene drugs (C60). Voyageur has developed five barium contrast products that have Health Canada licenses.
Voyageur's business plan is set to generate cash flow by partnering with established third-party GMP pharmaceutical manufacturers in Canada thereby ensuring the validation of its products by regulatory agencies worldwide. As Voyageur solidifies its presence in the market, it plans to transition into a high-margin domestic manufacturer of radiology drugs, further expanding its revenue streams.
At the core of its operations, Voyageur owns a 100% interest in the Frances Creek barium sulfate (barite) project. Currently, the world’s pharmaceutical barium sulfate is almost entirely synthetically produced which management believes results in a less effective imaging quality product. Voyageur’s Frances Creek resource boasts a rare and high grade mineral suitable for the pharmaceutical marketplace that Voyageur believes will replace the current synthetic products with higher quality lower cost imaging products.
Voyageur's ambitious vision is to become the first vertically integrated company in the radiology contrast media drug market. By controlling all primary input costs, from the sourcing of raw materials to final production, Voyageur intends to ensure quality and cost efficiency. With its approach, it embodies the motto of " From Earth to Bottle ," highlighting Voyageur's commitment to responsible sourcing and manufacturing practices.
For Further Information:
Neither the TSX Venture Exchange nor its Regulation Services Provider (as that term is defined in the policies of the TSX Venture Exchange) accepts responsibility for the adequacy or accuracy of this news release.
Cautionary Statement Regarding “Forward-Looking” Information
Copyright (c) 2026 TheNewswire - All rights reserved.
© 2026 Canjex Publishing Ltd. All rights reserved.