Toronto, Ontario--(Newsfile Corp. - March 2, 2026) - Theralase® Technologies Inc. (TSXV: TLT) (OTCQB: TLTFF) ("Theralase®" or the "Company"), a clinical stage pharmaceutical company dedicated to the research and development of energy-activated small molecules for the safe and effective destruction of cancer, bacteria and viruses, is pleased to announce that recent in vitro data demonstrates an enhanced bladder cancer cell kill, when light-activated Ruvidar® is combined with recombinant human interferon alpha-2b ("rhIFNα2b" or "interferon").
In preclinical research, T24 human bladder cancer cells were treated with two concentrations of light-activated Ruvidar® or left untreated. After activation with green light, cells were exposed to increasing doses of rhIFNα2b. The effectiveness of the treatment was assessed by how many cancer cells were killed 48 hours post-treatment.
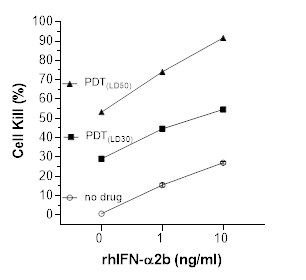
Figure 1: T24 human bladder cancer cell kill versus light-activated Ruvidar® and rhIFNα2b
To view an enhanced version of this graphic, please visit:
https://images.newsfilecorp.com/files/2786/285711_3645ee4124d61859_001full.jpg
Note: LD30 (Lethal Dose, 30%) is the amount of a substance required to kill 30% of a test population in a single dose, serving as a measure of acute toxicity. LD50 (Lethal Dose, 50%) is the amount of a substance required to kill 50% of a test population in a single dose.
Key findings include:
Light-activated Ruvidar® induced dose-dependent cytotoxicity in T24 human bladder cancer cells
rhIFNα2b induced dose-dependent cytotoxicity in T24 bladder cancer cells
The combination of light-activated Ruvidar® with rhIFNα2b resulted in 50 to 65% higher total cell kill compared to no light-activated Ruvidar®
Enhanced cytotoxic effects were observed across increasing concentrations of rhIFNα2b, suggesting a strong additive effect
Light-activated Ruvidar® works by directly killing tumor cells causing them to die in a way that helps the immune system recognize the cancer. This process releases tumor-specific signals that activate both the body's immediate (innate) and long-term (adaptive) immune responses.
Interferon alpha-2b works differently. It helps slow cancer cell growth, triggers cancer cell death and stimulates immune activity within the bladder to help eliminate bladder cancer cells.
The in vitro results suggest that the tumor reduction and immune activation induced by light-activated Ruvidar® may increase the sensitivity of residual tumour cells to interferon activity. This combined effect could potentially enhance treatment efficacy and extend the durability of response in patients with high-risk BCG-unresponsive NMIBC CIS.
These findings support the scientific rationale for combining Ruvidar® with interferon, which is planned in the Company's upcoming combinational clinical study, which will enroll and treat patients diagnosed with Bacillus Calmette-Guérin ("BCG")-Unresponsive Non-Muscle Invasive Bladder Cancer ("NMIBC") Carcinoma In Situ ("CIS") with or without resected papillary disease (±Ta/T1).
Mark Roufaiel, PhD, Research Scientist at Theralase® commented, "These findings provide mechanistic support for our combinational clinical study. By inducing targeted tumor destruction and immune activation with light-activated Ruvidar®, we hope to create a microenvironment more responsive to interferon alpha-2b. The enhanced total cell kill observed in vitro supports the potential of this complementary approach to improve outcomes for patients with BCG-unresponsive NMIBC."
Arkady Mandel, MD, PhD, DSc, Chief Scientific Officer, Theralase® stated, "Our preclinical research demonstrates that light-activated Ruvidar® combined with interferon, may provide significant benefit to patients afflicted with high-risk BCG-unresponsive NMIBC CIS, with or without papillary tumours. This combinational approach aligns with current thinking of combining world-class drugs to offer even higher efficacy, duration of response and safety for patients. I believe that the strong preclinical results demonstrated today will be confirmed clinically in the upcoming collaborative clinical study."
Roger DuMoulin-White, BSc, P.Eng, Pro.Dir, President, Chief Executive Officer and Chairman of the Board of Theralase®, added, "Our preclinical research has been used successfully to support design and development of our clinical programs. This has allowed our clinical programs to succeed, both in safety and efficacy, as they are firmly rooted in science. I look forward to reviewing the clinical data from the upcoming combinational clinical study."
About Ruvidar®:
Ruvidar® is a small molecule activated by energy, intended for the safe and effective destruction of cancer, bacteria and viruses.
About Theralase® Technologies Inc.:
Theralase® is a clinical stage pharmaceutical company dedicated to the research and development of energy-activated small molecules for the safe and effective destruction of cancer, bacteria and viruses.
Additional information is available at www.theralase.com and https://www.sedarplus.ca
Neither TSX Venture Exchange nor its Regulation Services Provider (as that term is defined in the policies of the TSX Venture Exchange) accepts responsibility for the adequacy or accuracy of this release.
Forward-Looking Statements
This news release contains Forward-Looking Statements ("FLS") within the meaning of applicable Canadian securities laws. Such statements include; but, are not limited to statements regarding the Company's proposed development plans with respect to small molecules and their drug formulations. FLS may be identified by the use of the words "may, "should", "will", "anticipates", "believes", "plans", "expects", "estimate", "potential for" and similar expressions; including, statements related to the current expectations of the Company's management regarding future research, development and commercialization of the Company's small molecules; their drug formulations; preclinical research; clinical studies and regulatory approvals.
These statements involve significant risks, uncertainties and assumptions; including, the ability of the Company to fund and secure regulatory approvals to successfully complete various clinical studies in a timely fashion and implement its development plans. Other risks include: the ability of the Company to successfully commercialize its small molecule and drug formulations; access to sufficient capital to fund the Company's operations is available on terms that are commercially favorable to the Company or at all; the Company's small molecule and formulations may not be effective against the diseases tested in its clinical studies; the Company fails to comply with the terms of license agreements with third parties and as a result loses the right to use key intellectual property in its business; the Company's ability to protect its intellectual property; the timing and success of submission, acceptance and approval of regulatory filings. Many of these factors that will determine actual results are beyond the Company's ability to control or predict.
Readers should not unduly rely on these FLS, which are not a guarantee of future performance. There can be no assurance that FLS will prove to be accurate as such FLS involve known and unknown risks, uncertainties and other factors, which may cause actual results or future events to differ materially from the FLS.
Although the FLS contained in the press release are based upon what management currently believes to be reasonable assumptions, the Company cannot assure prospective investors that actual results, performance or achievements will be consistent with these FLS.
All FLS are made as of the date hereof and are subject to change. Except as required by law, the Company assumes no obligation to update such FLS.
For investor information on the Company, please feel to reach out Investor Inquiries - Theralase Technologies.

To view the source version of this press release, please visit https://www.newsfilecorp.com/release/285711

© 2026 Canjex Publishing Ltd. All rights reserved.