- GLP-compliant program covering rodents, non-rodents, and primates will define safety, MTD and safe dose ranges, targeting weight loss and TIIDM
- Novel Ibogaine formulation development for mental health treatment including addiction
- Studies support upcoming regulatory submission and initiation of Phase I trial in Australia
Vancouver, British Columbia--(Newsfile Corp. - April 24, 2026) - SureNano Science Ltd. (CSE: SURE) (OTCQB: SURNF) ("SureNano" or the "Company"), parent company of GlucaPharm Inc. ("GlucaPharm"), a biopharmaceutical company developing novel therapeutic multi-receptor GLP-1 agonist peptide for the treatment of Type II Diabetes and Obesity candidates, today announced the planning of a comprehensive U.S. Food and Drug Administration (FDA) pathway Investigational New Drug (IND)-enabling Good Laboratory Practice (GLP) toxicology and pharmacology study program for its lead candidate GEP-44 (the "Program"). The Program is expected to be conducted in collaboration with a premier global Clinical Research Organization (CRO), LabCorp, known for its expertise in preclinical development, subject to entering into definitive collaboration agreements and the funding of related amounts.
This pivotal, GLP-compliant package is specifically designed to assess safety, toxicology, and pharmacology across multiple species, including rodents and non-rodent primates, to evaluate the safety and feasibility of the approach for human clinical application. The studies will provide comprehensive data to determine the maximum tolerated dose (MTD), identify potential biomarkers for monitoring toxicity, assess the reversibility of toxic effects, and establish the safety margin needed for first-in-human studies.
"Initiating these U.S. Food and Drug Administration (FDA) IND-enabling studies is a major milestone for SureNano Science and GlucaPharm, moving us firmly towards the final, rigorous evaluation in various clinical phases before entering the clinic," said Nihar Pandey, PhD., Chief Scientific Officer at SureNano. "Partnering with a globally renowned CRO like LabCorp will ensure our studies meet the highest regulatory standards. This package is vital to de-risk our lead candidate and is designed to support our strategy of launching a Phase I clinical trial in Australia."
Following the successful completion of these studies, SureNano intends to submit a Clinical Trial Notification (CTN) in Australia to begin U.S. Food and Drug Administration (FDA) Phase I testing in patients. Australia is chosen for its efficient regulatory framework, high-quality research infrastructure, and accelerated path to first-in-human studies.
With the progression of its lead asset GEP-44, a novel, patented peptide developed by researchers at Syracuse University, similar in function to Ozempic®, Wegovy®, Mounjaro®, Retatrutide targeting comparable or superior efficacy, improved patient tolerability and delivery potential, SureNano is positioning itself in a highly competitive and fast-growing therapeutic space where even incremental advances can have large commercial impact.
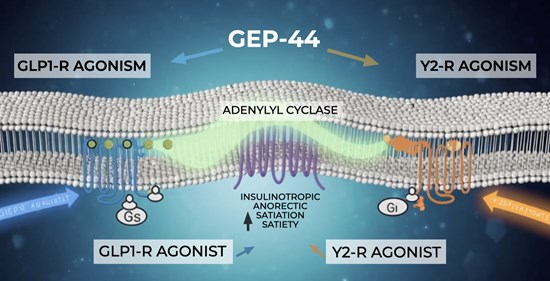
Figure 1: GEP44 Process Diagram
To view an enhanced version of this graphic, please visit:
https://images.newsfilecorp.com/files/8490/294091_65f03f2944a34af8_001full.jpg
Addition of Ibogaine Intellectual Property Opportunity to Company Portfolio
U.S. President Donald Trump recently signed an Executive Order designed to fast-track both research on and access to these psychedelic drugs with a focus on Ibogaine, including a $50 million investment in state governments to study how psychedelics might help people struggling with mental health illnesses.
The U.S. Food and Drug Administration (FDA) also issued Investigational New Drug (IND) clearance for Ibogaine to allow for human testing. Given this, we now have a clearance for Ibogaine, which will allow for human clinical trials for the first time in the United States, paving the way for more research and, ultimately, a development of commercial FDA approved treatment options using Ibogaine.
The Company believes Ibogaine and next-generation Ibogaine analogues have demonstrated potential in addressing addiction and mental health disorders, including opioid dependence and PTSD symptoms through mechanisms believed to involve modulation of multiple neurotransmitter systems and neuroplasticity pathways. President Donald Trump and super influencer Joe Rogan have commented on the merits of Ibogaine based treatments.
SureNano has initiated discussions with academic institutions and independent research groups to evaluate opportunities to license or acquire intellectual property related to the synthesis, formulation, and delivery of Ibogaine and structurally related compounds.
Discussions remain at an early stage, and there can be no assurance that any transaction or licensing agreement will be completed. The Company will provide further updates as material developments occur.
Investor Outreach & Awareness Campaign Agreement
The Company is also pleased to announce various strategic marketing and investor relations engagements (the "Engagements") with arms-length independent contractors and agencies, with the aim of developing the Company's communication strategy and strengthening exposure to a wider audience.
Emerging Growth, LLC
A media agency agreement dated April 22, 2026, has been executed by the Company with Emerging Growth, LLC doing business as TDM Financial ("TDM Financial") (the "TDM Financial Service Agreement"), a U.S.-based firm specializing in investor-focused marketing and communications. Under the terms of the TDM Financial Service Agreement, TDM Financial will execute a comprehensive investor outreach and awareness campaign over a six-month period which commenced on April 22, 2026. The campaign is designed to reach a broad audience of highly targeted investors across the United States and Canada through a combination of digital media channels and social platforms. Key elements of the campaign include distribution of sponsored, native-style content across leading financial websites and investor-focused social media platforms, including X (formerly Twitter), Reddit, TikTok, and LinkedIn, and the development and production of multimedia content, including videos, infographics, and editorial articles tailored to communicate the Company's corporate story and growth strategy. In accordance with the terms and conditions of the TDM Financial Service Agreement and as consideration for the services provided by TDM Financial, the Company has agreed to pay TDM Financial a total cash consideration of USD $26,667, payable in installments over the duration of the agreement. TDM Financial and its principals are arm's length from the Company and do not have any interest, direct or indirect, in the Company or its securities nor do they have any right or intent to acquire such an interest. TDM Financial's business is located at 600 E. 8th St., Whitefish, MT, USA 59937, and the contact information is as follows:
Tel: 1 (206) 369-7050
Website: https://www.tdmfinancial.com
Email: flane@cfnmedia.com
Investor Brand Network
A media agency agreement dated April 23, 2026, has been executed by the Company with Investor Brand Network doing business as IBN ("IBN") (the "IBN Service Agreement"), a multifaceted financial media and communications organization, to support the Company's investor awareness strategy. IBN will leverage its extensive syndication network, which includes over 5,000 media outlets, newsletters, social channels, and wire services, to expand the Company's reach to a broader U.S. investor audience. With more than 20 years of experience and a track record of serving over 600 public and private clients, IBN delivers comprehensive communications solutions through its network of financial brands and syndication partners. Under the terms of the agreement, IBN will provide corporate communication and awareness solutions to the Company over a six-month initial period commencing on May 4, 2026. IBN will assist SureNano in refining and strengthening its market awareness profile with the goal of maximizing shareholder value. In accordance with the terms and conditions of the IBN Service Agreement and as consideration for the services provided by IBN, the Company has agreed to pay IBN a quarterly cash consideration of USD $29,000, over the duration of the agreement, then quarter to quarter after the first six-month term, until cancellation. IBN and its principals are arm's length from the Company and do not have any interest, direct or indirect, in the Company or its securities nor do they have any right or intent to acquire such an interest. IBN's business is located at 1108 Lavaca Street Suite 110, Austin, Texas, USA, 78701 and the contact information is as follows:
Tel: 1 (512) 354-7000
Website: www.investorbrandnetwork.com
Email: Editor@InvestorBrandNetwork.com
The Market Link (iHub)
A media agency agreement dated April 23, 2026, has been executed by the Company with iHub Inc. DBA The Market Link ("TML"), a global department and brand, operated and owned entirely by TML, (the "TML Agreement"), pursuant to which TML has agreed to provide a marketing campaign, as part of which it will execute an enhanced news release distribution for the Company including news blast and forum boost advertising. The TML Service Agreement remains in effect until the retainer and inventory is depleted, with services commencing on April 24, 2026, the campaign period, and will not automatically renew. In accordance with the terms and conditions of the TML Service Agreement and as consideration for the services provided by TML, the Company has agreed to provide TML with a one-time cash fee of USD $10,000 plus applicable taxes. TML and its principals are arm's length from the Company and do not have any interest, direct or indirect, in the Company or its securities nor do they have any right or intent to acquire such an interest. TML's business is located at P.O. Box 780, Harrisonville, MO, 64701, USA, and the contact information is as follows:
Tel: 1 (778) 588-7012
Website: Investorshub.com
Email: support@advfn.com
Milky Way Marketing Inc. and Blue Sun Productions Inc. (BTV)
A service agreement dated February 23, 2026, has been executed by the Company with Milky Way Marketing Inc. and Blue Sun Productions Inc. ("BTV") (the "BTV Service Agreement"), a marketing and content agency. Pursuant to the terms and conditions of the BTV Service Agreement, BTV has agreed to provide the Company with a marketing and broadcast package that includes 30 second produced graphical style TV spot for broadcast, TV ad spots and forty (40) BNN Bloomberg broadcasts, along with a stock ticker ad and newsletter eblast. BTV will provide its services until the ads have been completed running or for a period of approximately 6 weeks, with ads to begin running on or around May 1, 2026. In accordance with the terms and conditions of the BTV Service Agreement and as consideration for the services provided by BTV, the Company has agreed to pay BTV a cash fee of $22,300 plus GST to be added where applicable. BTV and its principals are arm's length from the Company and do not have any interest, direct or indirect, in the Company or its securities nor do they have any right or intent to acquire such an interest. BTV's business is located at #17-19257B Enterprise Way, Surrey, BC, Canada, and the contact information is as follows:
Tel: 1 (604) 664-7401
Email: jessica@b-tv.com
Stock Options Grant
SureNano also announces that it has granted 1,500,000 stock options to various directors, officers, and consultants of the Company. The options will vest immediately and may be exercised at an exercise price of CAD $0.30 per common share, for a period of 3 years from the date of issuance or earlier in accordance with the Company's incentive stock option plan.
About SureNano Science Ltd.:
SureNano Science Ltd. (CSE: SURE) is a Canadian life sciences company focused on acquiring, developing, and advancing innovative pharmaceutical and biotechnology assets with the potential to address large and growing global health markets. SureNano Science Ltd. has acquired GlucaPharm Inc., a next-generation GLP-1 pharmaceutical company developing GEP44, a patented peptide targeting obesity and metabolic disorders with improved tolerability and potential non-injectable delivery (refer to press release dated February 23, 2026). The initial business of SureNano Science Ltd. is the sale and distribution of the SureNano™ surfactant, which is a ready-to-mix food grade compound that provides the base for high performance nanoemulsions to create incredibly homogeneous and stable products while maximizing bioavailability, clarity, and taste. The Company has an exclusive license to distribute the SureNanoTM surfactant within Canada; Oklahoma, USA; and Colorado, USA. SureNano Science Ltd. is now developing into a pharmaceutical focused company through the advancement of a patented therapeutic candidate designed to address obesity and metabolic disease.
Forward-Looking Information
This press release may include forward-looking information within the meaning of Canadian securities legislation, concerning the business of SureNano. Forward-looking information is based on certain key expectations and assumptions made by the management of SureNano. In some cases, you can identify forward-looking statements by the use of words such as "will," "may," "would," "expect," "intend," "plan," "seek," "anticipate," "believe," "estimate," "predict," "potential," "continue," "likely," "could" and variations of these terms and similar expressions, or the negative of these terms or similar expressions. Forward-looking statements in this press release include but are not limited to statements or information with respect to: funding for the Program; the entering into of definitive agreements related to the Program; the collaboration with LabCorp; the FDA pathway IND enabling GLP toxicology and pharmacology study program for GEP-44; future studies and programs involving the Company and its drug candidates; future drug trials, including the intended phase 1 clinical trial in Australia; the Company's growth and strategic initiatives; the Company's positioning among competitors; development milestones for the Company's drug candidates; the impact and results of the Company's drug candidates, including GEP44; the effectiveness and impact of Donald Trumps' executive order and the FDAs IND clearance; the effectiveness of Ibogaine in addressing substance use disorders; discussion regarding the Company and academic institutions and independent research groups regarding intellectual property; the TDM Financial Servies Agreement, IBN Service Agreement, and BTV Service Agreement.
Forward-looking information involves numerous risks and uncertainties, and actual results might differ materially from results suggested in any forward-looking information. These risks and uncertainties include, among other items: market prices, study and trial results, continued availability of capital and financing, and general economic, market, or business conditions. Although the Company believes that the expectations reflected in such forward-looking statements are reasonable, it can give no assurance that such expectations will prove to have been correct. The Company cautions that actual performance will be affected by a number of factors, most of which are beyond its control, and that future events and results may vary substantially from what the Company currently foresees. Factors that could cause actual results to differ materially from those in forward-looking statements include: expectations regarding the size of the markets the company operates in; the ability of the Company to successfully achieve its business objectives; plans for growth, political, social and environmental uncertainties; presence of laws and regulations which may impose restrictions in the markets where the Company operates; market volatility; fluctuations in investor sentiment; changes in the Company's business plans; and trial and study results. Accordingly, readers should not place undue reliance on forward-looking information and statements. Readers are cautioned that reliance on such information and statements may not be appropriate for other purposes.
The forward-looking statements are expressly qualified in their entirety by this cautionary statement. The information contained herein is stated as of the current date and is subject to change after that date. The Company does not assume the obligation to revise or update these forward-looking statements, except as may be required under applicable securities laws.
The Canadian Securities Exchange (CSE) has not reviewed and does not accept responsibility for the adequacy or the accuracy of the contents of this release.

To view the source version of this press release, please visit https://www.newsfilecorp.com/release/294091

© 2026 Canjex Publishing Ltd. All rights reserved.