/NOT FOR DISTRIBUTION TO UNITED STATES NEWSWIRE SERVICES OR FOR DISSEMINATION IN THE UNITED STATES/
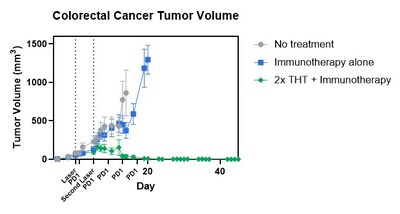
HALIFAX, NS, April 7, 2026 /CNW/ - Sona Nanotech Inc. (CSE: SONA) (OTCQB: SNANF) (the "Company", "Sona") is excited to announce publication of a preclinical study using its Targeted Hyperthermia Therapy ("THT") to treat cancer, in the Journal of Nanobiotechnology (https://bit.ly/47M2VoQ) which demonstrated treatment durability to the end of the 45 day study period when it was given in combination with immunotherapy. Building on its prior body of preclinical research treating melanoma and breast cancer (as published in Frontiers in Immunology), this study was conducted in an immunologically 'cold' colorectal cancer (CT26) tumor model. In this combination protocol, CT26 tumor-bearing animals were treated with two consecutive treatments of THT plus standard PD-1 inhibitor immunotherapy followed by three additional PD-1 treatments (the "Study"). Animals were followed for 45 days following initiation of treatment as per study protocol. At completion of the Study 38% of animals given the combined therapy were alive and disease free.
Sona Nanotech CEO, David Regan commented, "We've all recently heard the shocking news that colon cancer is now the leading cause of cancer death for adults under 50. This timely preclinical evidence of strong immune activation in colon cancer using our THT/immunotherapy combination protocol reaffirms our confidence in pursuing Sona's THT to spark immunity in typically immunotherapy resistant cancers. We are certainly proud of the quality of this peer-reviewed research and its acceptance in the highly respected Journal of Nanobiotechnology. This study, together with the safety and tolerability data from our recently completed, first-in-human study of THT as a monotherapy in melanoma, lays the foundation for our next trial that will combine our THT with immunotherapy."
Study principal investigator, lead author and Sona Chief Medical Officer, Dr. Carman Giacomantonio, commented, "We have focused our preclinical research on immunogenically cold cancers i.e. cancers that do not respond to today's standard-of-care immunotherapy, to prove our hypothesis that Sona's THT activates immunity. This is the third preclinical cancer-type in which we have confirmed our hypothesis, and our first publication delving deeply into the biology of our treatments. In this study, where no animals responded to a standard immunotherapy alone, 100% of animals in the THT treatment group responded to that same immunotherapy. This relates incredibly favorably to the less than ~15% success rate of immunotherapy when used to treat colorectal cancer in most humans due to low immunogenicity, giving us tremendous hope for its ability to change lives if translated successfully into a human therapy. Importantly, 38% (8 of 21) of those tumors completely cleared by day 24 following initiation of treatment and remained cancer-free to the end of the study. This is remarkably fast by any standard of measure, and these important findings will inform the protocol for our next clinical study."
About Sona Nanotech Inc.
Sona Nanotech, a nanotechnology life sciences company, is developing Targeted Hyperthermia™, a photothermal cancer therapy, which uses therapeutic heat to treat solid cancer tumors. The heat is delivered to tumors by infrared light that is absorbed by Sona's gold nanorods in the tumor and re-emitted as heat. Therapeutic heat (42-48°C) stimulates the immune system, shrinks tumors, inactivates cancer stem cells, and increases tumor perfusion – thus enabling drugs to reach all tumor compartments more effectively. The size, shape, and surface chemistry of the nanorods target the leaky vasculature of solid tumors, and the selective thermal sensitivity of tumor tissue enables the therapy to deliver clean margins. Targeted Hyperthermia promises to be safe, effective, minimally invasive, competitive in cost, and a valuable adjunct to drug therapy and other cancer treatments.
Sona has developed multiple proprietary methods for the manufacture of gold nanoparticles which it uses for the development of both cancer therapies and diagnostic testing platforms. Sona Nanotech's gold nanorod particles are cetyltrimethylammonium ("CTAB") free, eliminating the toxicity risks associated with the use of other gold nanorod technologies in medical applications. It is expected that Sona's gold nanotechnologies may be adapted for use in applications, as a safe and effective delivery system for multiple medical treatments, subject to the approval of various regulatory boards, including Health Canada and the FDA.
CAUTIONARY STATEMENT REGARDING FORWARD-LOOKING INFORMATION: This press release includes certain "forward-looking statements" under applicable Canadian securities legislation, including statements regarding the anticipated applications and potential opportunities of Targeted Hyperthermia Therapy, Sona's preclinical and clinical study plans, future patent filings and its product development plans. Forward-looking statements are necessarily based upon a number of assumptions or estimates that, while considered reasonable, are subject to known and unknown risks, uncertainties, and other factors which may cause the actual results and future events to differ materially from those expressed or implied by such forward-looking statements, including the risk that Sona may not be able to successfully obtain sufficient clinical and other data to submit regulatory submissions, raise sufficient additional capital, secure patents or develop the envisioned therapy, and the risk that THT may not prove to have the benefits currently anticipated. There can be no assurance that such statements will prove to be accurate, as actual results and future events could differ materially from those anticipated in such statements. Accordingly, readers should not place undue reliance on forward-looking statements. Sona disclaims any intention or obligation to update or revise any forward-looking statements, whether as a result of new information, future events or otherwise, except as required by law.
SOURCE Sona Nanotech Inc.

 View original content to download multimedia: http://www.newswire.ca/en/releases/archive/April2026/07/c1362.html
View original content to download multimedia: http://www.newswire.ca/en/releases/archive/April2026/07/c1362.html