- Positive Full Results from Phase 1a Trial of Anti-TSLP/IL-4Rα Bispecific Antibody ATI-052 Exceed Aclaris’ Target Profile, Validating Potential Best-in-Class Potency Advantage and Opportunity for Extended Dosing -
- Estimated Half-Life of Approximately 45 Days; Unlocks Opportunity for up to Three-Month Dosing Interval -
- Complete and Sustained Inhibition of TSLP-Induced and IL-4 Induced CCL17 (TARC) Provides Potential to Raise Efficacy Ceiling in Th2-Driven Diseases -
- Enrollment and Dosing Ongoing in Phase 1b Trials in Asthma and Atopic Dermatitis (AD) with Top Line Results Expected in the Second Half of 2026 -
- Lichen Planus Selected as Lead Indication for ATI-2138; Potential to be First Mechanistically Complete Therapeutic Candidate Designed to Address Root Inflammation and Symptoms -
- Management to Host a Conference Call to Discuss Update Today at 8:30 AM EST -
WAYNE, Pa., April 28, 2026 (GLOBE NEWSWIRE) -- Aclaris Therapeutics, Inc. (NASDAQ: ACRS), a clinical-stage biopharmaceutical company focused on developing novel product candidates for immuno-inflammatory diseases, today provided a clinical update on its biologic and oral inhibitor compounds including positive full top line results from the first-in-human Phase 1a single (SAD) and multiple ascending dose (MAD) trial of its anti-TSLP/IL-4Rα bispecific antibody ATI-052 and the selection of lichen planus (LP) as the lead indication for its selective ITK/JAK3 inhibitor ATI-2138.
“These results validate that ATI-052 is a highly potent and well tolerated bispecific antibody that has the potential to be highly effective in a variety of inflammatory and immunological disorders,” said Dr. Neal Walker, Chief Executive Officer of Aclaris. “Based on the strong safety and tolerability profile and dose proportional pharmacokinetic and pharmacodynamic profiles that support the potential for an extended dosing schedule of up to every three months and the potential for synergistic efficacy resulting from its dual inhibition of TSLP and IL-4Rα, we are rapidly progressing this molecule through two proof-of-concept studies and plan to initiate a Phase 2b program in the fourth quarter of 2026.”
Continued Dr. Walker, “We also intend to initiate a phased multi-part Phase 2b basket study in lichen planus with ATI-2138 later this year. ATI-2138 is the only known drug that hits pathogenic T cells, the key drivers of interface dermatitis disorders like LP, by inhibiting both TCR and effector cytokine mediated activation - and as such we believe it is an ideal mechanistic fit for this indication. LP causes debilitating symptoms and significant impacts to quality of life across the spectrum of subtypes; there are no therapeutic options approved for use in this disease. We expect our Phase 2b program to initially focus on erosive mucosal and cutaneous LP, starting in the second half of this year, and move into lichen planopilaris shortly thereafter.”
ATI-052: Anti-TSLP/IL-4Rα Bispecific Antibody
Full Top Line Phase 1a SAD/MAD Results
ATI-052 is an investigational anti-TSLP and anti-IL-4Rα bispecific antibody that exhibits high binding affinity to, and dual blockade of, both the TSLP receptor signal transduction and downstream IL-4Rα activation thereby inhibiting this central proinflammatory pathway. By addressing the shared receptor IL-4Rα, ATI-052 blocks IL-4 and IL-13 biological activity, key cytokines driving Th2 inflammation. The Company provided interim results in January 2026 (press release here). Today’s update provides full top line results including:
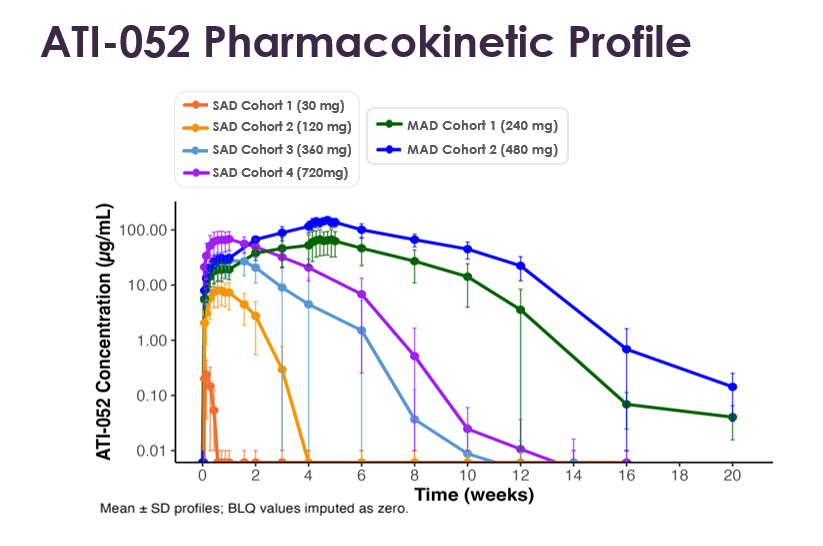
- ATI-052 exhibited a potential best-in-class pharmacokinetic (PK) profile, including an estimated half-life of approximately 45 days1. Dose proportional PK was observed across the pharmacologic dose range, including dose proportional increases in Cmax (maximum peak concentration) and AUC (area under the curve; a measure representing total systemic exposure).
- Pharmacodynamic (PD) results validate the potency of ATI-052, including robust target engagement and near complete target occupancy. ATI-052 demonstrated complete and sustained inhibition through at least week 20 (four months post last dose) of ex vivo TSLP stimulated CCL17/TARC and at least week 12 (two months post last dose) of ex vivo IL-4 stimulated CCL17/TARC in the 480 mg MAD cohort.
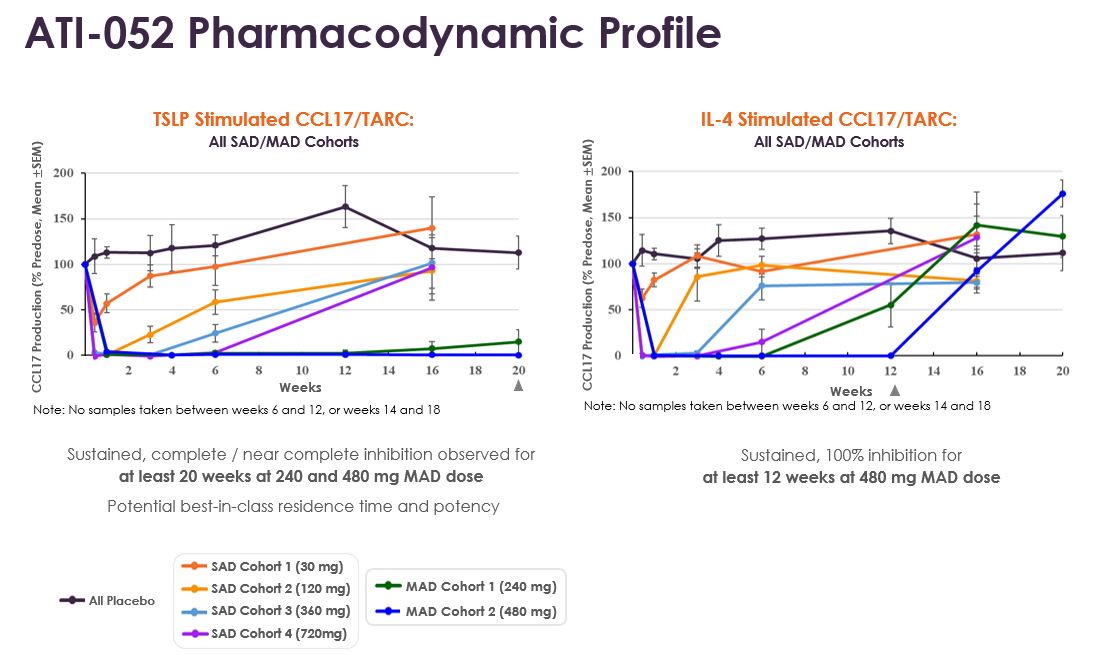
- The combination of the strong and sustained PK duration and PD effect supports the potential for up to every three-month dosing.
- No impact of anti-drug antibodies (ADA) on PK or PD has been observed in this trial.
- ATI-052 was well tolerated and demonstrated a favorable safety profile across all SAD and MAD cohorts, consistent with the interim results; no safety signals including conjunctivitis were observed.
Phase 1a SAD/MAD Trial Design
The randomized, blinded, placebo-controlled Phase 1a portion of the first-in-human study was designed to evaluate the safety, tolerability, PK, and PD of subcutaneously administered ATI-052 in healthy adults receiving SAD and MAD doses. In the SAD portion, four cohorts of 8 healthy volunteers each were randomized 3:1 to receive a single dose of ATI-052 (30, 120, 360, or 720 mg) or placebo. In the MAD portion, two cohorts of 8 healthy volunteers each were randomized 3:1 to receive five doses of two dose levels of ATI-052 (240 or 480 mg) or placebo administered every 7 days. Participants were followed for safety for approximately 16 weeks for the SAD cohorts and 20 weeks for the MAD cohorts.
Ongoing and Future Clinical Trials
In January 2026, Aclaris announced the initiation of a Phase 1b proof-of-concept (POC) trial in patients with atopic dermatitis (EASI >21). In February 2026, the Company initiated a Phase 1b POC trial in patients with asthma on GINA (Global Initiative for Asthma) steps 2-4 treatment prior to screening. Enrollment is ongoing in both trials, and top line results from both are expected in the second half of 2026.
Given the results observed to date and its mechanism of action targeting TSLP as well as effects of IL-4 and IL-13 through blockade of the shared receptor IL-4Rα, the Company announced its intent to initiate a Phase 2b program with ATI-052, initially targeting asthma, in the fourth quarter of 2026.
ATI-2138: Selective ITK/JAK3 Inhibitor
ATI-2138 is a highly potent and selective novel investigational pharmacologic agent that acts as a dual inhibitor of interleukin-2-inducible T cell kinase (ITK) and Janus kinase 3 (JAK3); the dual mechanism enables deep suppression of pathogenic T-cells via inhibition of TCR signaling and effector cytokine activation.
Aclaris intends to initiate a phased multi-part Phase 2b basket study of ATI-2138 in lichen planus (LP), an unaddressed chronic, inflammatory, CD8+/Th1-driven interface dermatitis impacting approximately 0.1% to 1.0% of the population. The trial is expected to comprise the three most common LP subtypes: erosive mucosal, cutaneous, and lichen planopilaris (LPP), a rare form of LP that causes permanent hair loss. The most common symptoms of LP include sores, pain, difficulty eating, severe itch, scales/plaques, hair loss, and fatigue; quality of life is also affected in most patients, including due to anxiety and depression. There is also the potential for malignancy in certain subtypes. Disease management has focused on immunosuppression and symptom control; there are currently no FDA-approved therapies. Despite the lack of therapeutic options, approximately 40% of patients seek treatment. An oral agent like ATI-2138 has the potential to provide rapid symptom and itch relief and address multi-site (cutaneous, oral, and scalp) disease involvement. The Company estimates that the potential market opportunity in the U.S. for an oral therapeutic for LP exceeds $1.0 billion with opportunity up to $4.0 billion. Aclaris expects to initiate Part A (erosive mucosal; cutaneous) of this trial in the second half of 2026 and intends to move into Part B (LPP) soon thereafter.
“Lichen planus remains a challenging, immune-mediated disease with significant patient burden, and there is a clear need for additional therapies that can more effectively address both its symptoms and underlying pathology,” said Dr. Johann Gudjonsson, MD, PhD, Arthur C. Curtis Professor of Skin Molecular Immunology, University of Michigan Skin Research Center.
Webcast and Conference Call
Aclaris will host a webcast and conference call with slides today at 8:30 AM ET to discuss the full ATI-052 Phase 1a SAD/MAD results and an overview of the lead indication and upcoming clinical program for ATI-2138. The live and archived webcast will be available on the Events page of the Company’s website: https://investor.aclaristx.com/events. The webcast will be archived on the same page for 30 days following the event. If you would rather access the call via telephone: To register and receive a dial in number and unique PIN to access the live conference call, please follow this link to register online. Upon registering you will receive the dial-in info and a unique PIN to join the call as well as an email confirmation with the details.
About ATI-052
ATI-052 is an investigational humanized anti-TSLP and anti-IL-4Rα bispecific antibody that exhibits high binding affinity to and dual blockade of both the upstream thymic stromal lymphopoietin (TSLP) receptor signal transduction and downstream interleukin-4 receptor (IL-4R) activation thereby inhibiting this central proinflammatory pathway. ATI-052 targets TSLP, which sits at the top of the inflammatory cascade; by targeting IL-4Rα, it blocks both downstream IL-4 and IL-13, which are key cytokines involved in Th2-mediated inflammation and allergic diseases. ATI-052 exhibits potential best-in-class potency and utilizes the same TSLP antigen-binding fragment (Fab) region as bosakitug (ATI-045), retaining the dissociation kinetics, long residence time, and high potency advantages over comparator antibodies, but is engineered to bind more tightly to the neonatal Fc receptor (FcRn), potentially extending its half-life. ATI-052 has the potential to treat a variety of atopic, immunologic and respiratory diseases. Aclaris has the exclusive worldwide rights to ATI-052, excluding Greater China.
About ATI-2138
ATI-2138 is a highly potent and selective novel investigational pharmacologic agent that acts as a dual inhibitor of interleukin-2-inducible T cell kinase (ITK) and Janus kinase 3 (JAK3). ITK regulates T cell receptor signal transduction and inhibition of this kinase can affect T cell differentiation and activation. JAK3 is a key signal transduction kinase that forms a heterodimer with JAK1, modulates JAK1 phosphorylation of signal transducer and activator of transcription 5 (STAT5), and regulates cytokines that signal through the IL-2Rγc to affect lymphocyte proliferation and activation. The efficacy results exhibited in preclinical animal models of inflammation and autoimmune diseases, coupled with the favorable safety, pharmacokinetics, and pharmacodynamics profile observed in healthy human SAD and MAD studies and a Phase 2a trial in atopic dermatitis, support the potential for ATI-2138 to affect several human inflammatory diseases and further investigation of this molecule in patients with atopic and autoimmune diseases that are dependent on T cell function and/or IL-2Rγc signaling.
About Aclaris Therapeutics, Inc.
Aclaris Therapeutics, Inc. is a clinical-stage biopharmaceutical company developing a pipeline of novel product candidates to address the needs of patients with immuno-inflammatory diseases who lack satisfactory treatment options. The company has a multi-stage portfolio of product candidates powered by a robust R&D engine. For additional information, please visit www.aclaristx.com and follow Aclaris on LinkedIn.
Cautionary Note Regarding Forward-Looking Statements
Any statements contained in this press release that do not describe historical facts may constitute forward-looking statements as that term is defined in the Private Securities Litigation Reform Act of 1995. These statements may be identified by words such as “anticipate,” “believe,” “expect,” “intend,” “may,” “plan,” “potential,” “will,” and similar expressions, and are based on Aclaris’ current beliefs and expectations. These forward-looking statements include expectations regarding its development plans for ATI-052 and ATI-2138, including the timing to report results from its Phase 1b trials of ATI-052 in asthma and atopic dermatitis, the timing to initiate a Phase 2b trial of ATI-052 in asthma, and the timing and plans to initiate a Phase 2b basket study of ATI-2138 in LP, the therapeutic potential for ATI-052 and ATI-2138, including the potential for ATI-052 to be first- and best-in-class, and the potential for ATI-052 to support dosing of up to three months, and the potential for ATI-052 to treat atopic, immunologic, and respiratory diseases and ATI-2138 to treat atopic and autoimmune diseases. These statements involve risks and uncertainties that could cause actual results to differ materially from those reflected in such statements. Risks and uncertainties that may cause actual results to differ materially include uncertainties inherent in the conduct of clinical trials, risks associated with interim, topline and preliminary data, Aclaris’ reliance on third parties over which it may not always have full control, Aclaris’ ability to enter into strategic partnerships on commercially reasonable terms, the uncertainty regarding the macroeconomic environment and other risks and uncertainties that are described in the Risk Factors section of Aclaris’ Annual Report on Form 10-K for the year ended December 31, 2025, and other filings Aclaris makes with the U.S. Securities and Exchange Commission from time to time. These documents are available under the “SEC Filings” page of the “Investors” section of Aclaris’ website at www.aclaristx.com. Any forward-looking statements speak only as of the date of this press release and are based on information available to Aclaris as of the date of this release, and Aclaris assumes no obligation to, and does not intend to, update any forward-looking statements, whether as a result of new information, future events or otherwise.
1Based on accumulation ratio at 240 mg weekly dosing
Aclaris Therapeutics Contact:
Will Roberts
Senior Vice President
Corporate Communications and Investor Relations
(484) 329-2125
wroberts@aclaristx.com
Photos accompanying this announcement are available at
https://www.globenewswire.com/NewsRoom/AttachmentNg/6b2f9dba-280e-4444-8055-6f8112b06aa5
https://www.globenewswire.com/NewsRoom/AttachmentNg/fb3bc6d2-b0de-4917-843c-242f7f16a347



ATI-052 PK
ATI-052 Pharmacokinetic Profile
ATI-052 PD
ATI-052 Pharmacodynamic Profile
© 2026 Canjex Publishing Ltd. All rights reserved.