Toronto, Ontario--(Newsfile Corp. - April 13, 2026) - Theralase® Technologies Inc. (TSXV: TLT) (OTCQB: TLTFF) ("Theralase®" or the "Company"), a clinical stage pharmaceutical company dedicated to the research and development of energy-activated small molecules for the safe and effective destruction of cancer, bacteria and viruses has demonstrated a complete response of all animals treated with X-Ray-activated Rutherrin® in a preclinical animal model of Muscle Invasive Bladder Cancer ("MIBC").
Kaplan–Meier survival analysis from the orthotopic MIBC animal study demonstrated that 100% of the animals treated with X-Ray-activated Rutherrin® exhibited a complete response and remained cancer-free at the end of the study; whereas, animals assigned to the radiation only or untreated control group did not respond as well. (See Figure 1).
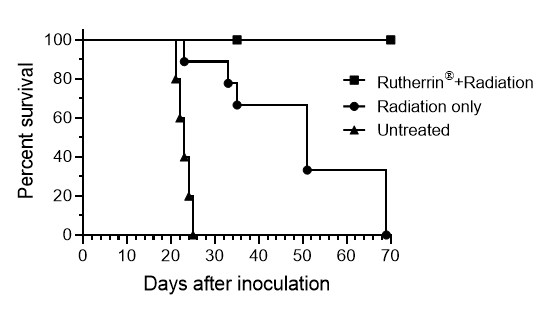
Figure 1: Kaplan-Meier Survival Curve Demonstrating Complete Response of Animals Treated with X-Ray-activated Rutherrin® in MIBC Animal Model
To view an enhanced version of this graphic, please visit:
https://images.newsfilecorp.com/files/2786/292023_8bea78c6048248b8_001full.jpg
The radiation only group demonstrated partial therapeutic benefit, with a progressive decline in survival over time. Animals in this group perished from the disease within 25 and 70 days.
Untreated animals in the control group perished from the disease between 20 and 23 days, consistent with aggressive tumor progression.
These findings build upon previously reported results1, now demonstrating complete response and the duration of that response in animals treated with X-Ray-activated Rutherrin®. The addition of Kaplan–Meier survival data demonstrates 100% survival of animals, treated with X-Ray-activated Rutherrin®, laying the groundwork for future clinical development.
Mark Roufaiel, PhD, Research Scientist at Theralase®, commented, "The latest survival data represents a key milestone in our preclinical program. While earlier results demonstrated a trend to complete tumour clearance, the Kaplan–Meier analysis confirms that this translates into a meaningful and durable survival benefit. These findings further support the potential of X-Ray-activated Rutherrin® versus radiation only to provide complete durable responses in aggressive cancers such as MIBC."
Arkady Mandel, MD, PhD, DSc, Chief Scientific Officer, Theralase®, stated, "Rutherrin® combined with low-dose radiation significantly improved survival in a preclinical MIBC mouse model. These results suggest that Rutherrin® may have clinical potential for locally advanced, muscle-invasive bladder cancer, where tumours invade the bladder’s muscle wall. Annually, up to 21,200 Americans and up to 184,000 people globally are diagnosed with MIBC.2,3 The standard of care predominantly involves either radical cystectomy or trimodal therapy (surgery, chemotherapy and radiation), yet 20 to 40% of patients still develop new pelvic tumors within two to three years, after treatment.4,5 Combining Rutherrin® with radiation or chemoradiotherapy may offer an effective, bladder-sparing approach to preserve function and enhance quality of life."
Roger DuMoulin-White, BSc, PEng, Pro.Dir, President, Chief Executive Officer and Chairman of the Board of Theralase®, added, "X-ray-activated Rutherrin® continues to impress all of us in terms of safety and efficacy in the destruction of a wide variety of cancerous tumours. Complete and durable response in a MIBC animal model is only the latest of its accolades. Pending GLP toxicology analysis, Theralase® plans to commence a Phase 0/I/II adaptive clinical study evaluating X-Ray-activated Rutherrin®, with or without surgery, chemotherapy and/or immunotherapy, subject to regulatory approval, in patients diagnosed with MIBC, in 2026."
References
1 Press Release. Theralase® Demonstrates That X-Ray-Activated Rutherrin® Is Significantly More Effective than Radiation Alone in the Destruction of Muscle Invasive Bladder Cancer. March 9, 2026
2 Key Statistics for Bladder Cancer. American Cancer Society. 2026
3 Li H et al. Regional, and National Burden and Trends of Bladder Cancer in Individuals Aged 55 Years and Older from 1990 to 2021: Findings from the Global Burden of Disease Study 2021. Int J Surg. 2025 Dec 1;111(12):8916-8933.
4 Eapen LJ et al. Enumerating pelvic recurrence following radical cystectomy for bladder cancer: A Canadian multi-institutional study. Can Urol Assoc J. 2016 Mar-Apr;10(3-4):90-4.
5 Damaj N et al. Management of bladder cancer recurrence following the trimodality therapy. Front Oncol. 2025
About Theralase® Technologies Inc.:
Theralase® is a clinical stage pharmaceutical company dedicated to the research and development of energy-activated small molecules for the safe and effective destruction of cancer, bacteria and viruses.
Additional information is available at www.theralase.com and www.sedarplus.ca.
Neither TSX Venture Exchange nor its Regulation Services Provider (as that term is defined in the policies of the TSX Venture Exchange) accepts responsibility for the adequacy or accuracy of this release.
Forward-Looking Statements
This news release contains Forward-Looking Statements ("FLS") within the meaning of applicable Canadian securities laws. Such statements include; but, are not limited to statements regarding the Company's proposed development plans with respect to small molecules and their drug formulations. FLS may be identified by the use of the words "may, "should", "will", "anticipates", "believes", "plans", "expects", "estimate", "potential for" and similar expressions; including, statements related to the current expectations of the Company's management regarding future research, development and commercialization of the Company's small molecules; their drug formulations; preclinical research; clinical studies and regulatory approvals.
These statements involve significant risks, uncertainties and assumptions; including, the ability of the Company to fund and secure regulatory approvals to successfully complete various clinical studies in a timely fashion and implement its development plans. Other risks include: the ability of the Company to successfully commercialize its small molecule and drug formulations; access to sufficient capital to fund the Company's operations is available on terms that are commercially favorable to the Company or at all; the Company's small molecule and formulations may not be effective against the diseases tested in its clinical studies; the Company fails to comply with the terms of license agreements with third parties and as a result loses the right to use key intellectual property in its business; the Company's ability to protect its intellectual property; the timing and success of submission, acceptance and approval of regulatory filings. Many of these factors that will determine actual results are beyond the Company's ability to control or predict.
Readers should not unduly rely on these FLS, which are not a guarantee of future performance. There can be no assurance that FLS will prove to be accurate as such FLS involve known and unknown risks, uncertainties and other factors which may cause actual results or future events to differ materially from the FLS.
Although the FLS contained in the press release are based upon what management currently believes to be reasonable assumptions, the Company cannot assure prospective investors that actual results, performance or achievements will be consistent with these FLS.
All FLS are made as of the date hereof and are subject to change. Except as required by law, the Company assumes no obligation to update such FLS.
For investor information on the Company, please feel to reach out Investor Inquiries - Theralase Technologies.

To view the source version of this press release, please visit https://www.newsfilecorp.com/release/292023

© 2026 Canjex Publishing Ltd. All rights reserved.